TubeClear News
Check out what's new for the TubeClear system.

Latest News
Partnership with PSU Nursing School
Actuated Medical's partnership with Penn State's College of nursing helped speed TubeClear to market and ensure clinician adoption. Janice Penrod, director of the Center for Nursing Research at Penn State’s College of Nursing commented, “It’s a perfect case of...
More News

Prophylactic use of the TubeClear system reduces feeding tube material buildup and enables optimal enteral nutrition delivery.
Enteral nutrition (EN) is provided for patients that have a functioning lower gastrointestinal (GI) tract but are unable to orally ingest nutrients and medication and are at risk for malnutrition.[1] Feeding tubes (Tubes) provide these patients essential nutrition,...
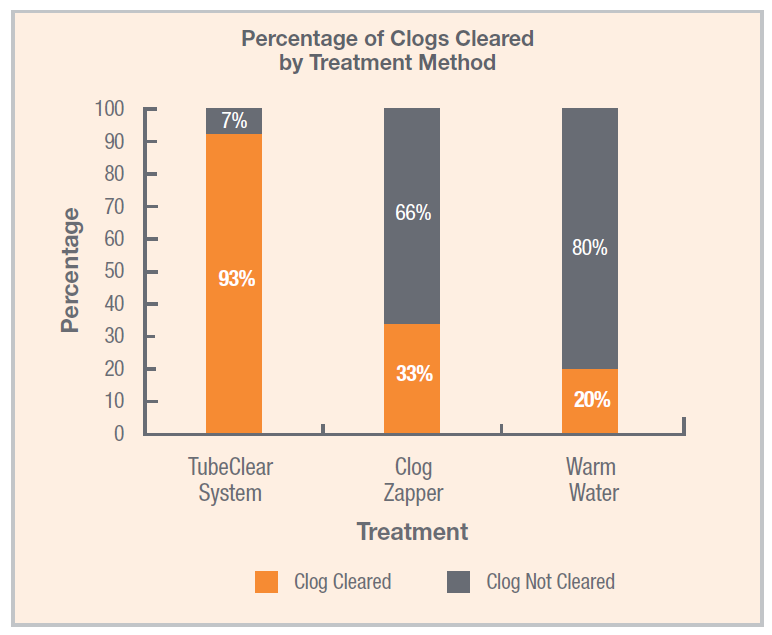
TubeClear System More Effective at Clearing Clogged Feeding Tubes
In a recent study published in Nutrition in Clinical Practice by Christopher M. Garrison, Ph.D., RN, CNE, the TubeClear system was found to be significantly more effective at removing clogs from feeding tubes than water or enzyme-based clog-clearing treatments.[1]...
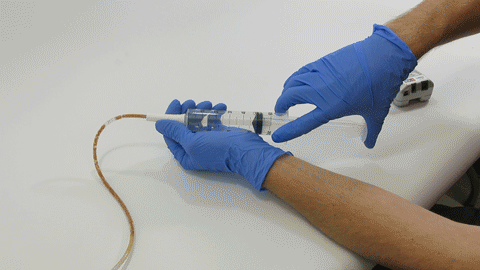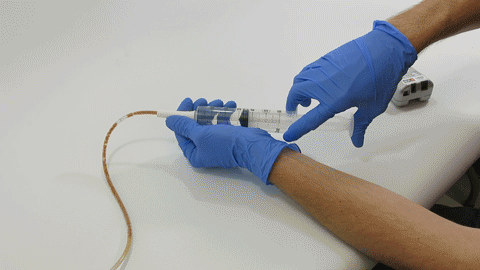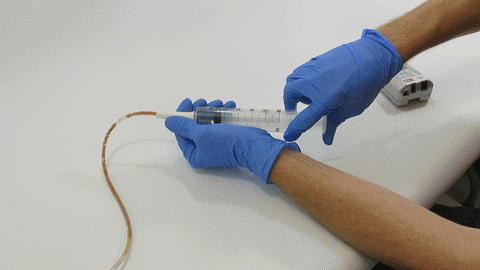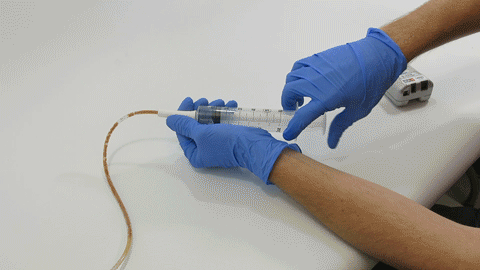
Syringe Force on Clogged Feeding Tubes
Many people think feeding tubes are merely used to deliver nutrition (i.e., feeding formula), however, they deliver life-sustaining medications too - it’s no wonder they tend to clog. At Actuated Medical, Inc., we developed the TubeClear System to keep enteral therapy...

The Impact of Clogged Feeding Tubes
Exhibiting the TubeClear System at the Oley Foundation’s Annual Conference was a new experience for the Actuated Medical team. While many conferences and trade shows target a specific audience, this one is a little different. A non-profit organization providing support to patients on total parenteral nutrition (TPN) or enteral nutrition (EN) and their families, the Oley Foundation invites everyone associated with these two nutritional health requirements to its Annual Conference. Attendees ranged from gastroenterologists and dietitians to the tube-fed patients themselves, allowing a glimpse at the bigger picture of EN and the impact of clogs. These are patients who rely on TPN or EN for their nutrition, medication, and hydration needs as they battle numerous conditions. For them, that tube is their lifeline – their world.
It was a humbling experience, to say the least. These individuals know feeding tubes, and they know clogs. In literature, clogs occur as often as 35% of the time. It seems like a small problem. But for the individual who experiences one, it’s a problem that can grow and affect many aspects of their lives.
Bench Top Studies
TubeClear System Clears Clogged NE Tubes: Benchtop Study
Small bore feeding tubes are used to provide essential nutrition and medication to patients at risk of malnutrition and dehydration due to an inability to ingest orally (1). Clogging is one of the most frequent mechanical complications of feeding tubes (2,3). Tubes...